1 Context
The COVID-19 pandemic has highlighted the need for dynamic and strategic preparedness to combat emerging epidemics and pandemic threats. Behavioural changes, climate changes, and an increasingly globalised world has led to an elevated risk for global health emergencies. Therefore, there is an urgent need to build a sustainable and resilient defence mechanism towards future epidemics and pandemics. One of the lessons learnt from COVID-19 is that rapid development and deployment of safe and effective vaccines is a cornerstone in containing a global pandemic. To achieve this, trustworthy and adequate information about vaccines clinical trials is essential to rapidly mobilise enrolment in clinical trials to test new vaccines and people for vaccination campaigns.
The European vaccine trial network, VACCELERATE ( https://www.vaccelerate.eu/ ), was set up to coordinate phases II and III COVID-19 vaccine trials across Europe. VACCELERATE’s goal is to become a single-entry point for multi-country vaccine trials in Europe, and to help address the need for greater inclusion of trial participants from populations under-represented in vaccine trials. “Underserved communities” is a term that usually refers to population groups that face barriers and challenges in accessing and using resources due to geographic location, religion, sexual orientation, gender identity, race, and ethnicity. Underserved populations usually encounter unique challenges, such as language and cultural barriers, physical and/or cognitive ability, alienage status, or age [IGI Global, n.d. ] Including underrepresented groups in clinical research is important as the results of a new intervention may not be otherwise generalised to a larger population, as well as those who stand to benefit the most. Logistics, social, psychological, and biological variations may all play a role in the outcome of vaccine trials. Thus, the inclusion of diverse groups of people will make it possible to find the balance of risk and benefits for different sub-groups.
Effective and targeted communication about vaccines and vaccine trials is an important foundation to ensuring high vaccination rates in general as well as achieving diversity and inclusiveness in clinical trial participation. A lack of dedicated information from official and reliable sources could further amplify the underrepresentation of particular population groups [Fazel et al., 2021 ]. For example, transgender people on hormone therapy may have concerns about taking a vaccine and the hormone therapy at the same time [Garg et al., 2021 ]. People experiencing homelessness, including those who are immigrants, may have concerns about vaccination as well. Teenagers may not be well informed about the principles of clinical trials [Fazel et al., 2021 ; Longchamps et al., 2021 ; Scherer et al., 2021 ]. The impact of communication will depend on a number of other factors, and crucially a well-developed health care system, health literacy in the population, knowledge accessibility, existing attitudes and trust in vaccines and the country’s health authorities. Nevertheless, effective and targeted communication remains an important factor in reaching high vaccine coverage and clinical trials participation in all groups.
In this study, we have compiled an inventory of online resources of information materials about COVID-19 vaccines and vaccine trials from official regional, national, and pan-European sources. Information about vaccine uptake and also specifically about participation in vaccine clinical trials was collected. Even though, COVID-19 vaccines have already been approved and administered to a large number of individuals, clinical studies have continued after the vaccines have been approved and marketed. Such “post-marketing clinical trials” aim to test the vaccines in additional population groups (e.g., children or pregnant women). We describe how the inventory was established and undertake a quantitative analysis to map the availability of public information about COVID-19 vaccines and vaccine trials in European countries and identify any information gaps. Furthermore, we also analysed any correlation between the amount of online information material and vaccination uptake in different European countries.
2 Methods
2.1 Collection of materials
Publicly available online communication materials (e.g., digital flyers, videos, audios, etc.) about COVID-19 vaccine and vaccine trials in Europe, clinical study participation, clinical trials, COVID-19 vaccines, and vaccine research and development (R&D) was compiled from official online sources of regional and national governments (e.g., ministries and public health authorities) across all 27 Member States (MS) of the European Union (EU), as well as Iceland, Israel, Norway, Serbia, Switzerland, Turkey and the United Kingdom (U.K.). Information was also collected from European institutions websites such as the European Centre for Disease Control (ECDC) and the European Commission and non-governmental organisations (NGOs), including patient organisations. To identify national government information and national public health authorities, the search initially relied on the trusted sources made available by the European Vaccination Information Portal ( https://vaccination-info.eu/en/trusted-sources ).
Additional sources/institutions were subsequently added through snowballing from the initial sources or directed searches on Google. The latter was verified to ensure their trustworthiness, specifically by confirming that the authors of the pages/portals were regional, national, or European governmental agencies, credible NGOs and/or other official organisations (e.g., public institutes, universities). Any organisation not recognised as an official public institution was excluded. All types of communication materials (e.g., digital flyers, videos, audios, etc.) available from the websites and portals of the various institutions were gathered. A cursory examination of each material’s content, keywords, title and summary was performed to categorise the collected material. Some communication channels were specifically excluded from further analysis, notably radio, television, and newspapers. Material collection took place between May and July 2021.
2.2 Definitions and categories
The information gathered was classified according to five main categories, namely 1) Media type, 2) Topic, 3) Target audience, 4) Country (the provenance of the institution that produced the media), and 5) Language.
2.2.1 Types of media
Eight different media (i.e., communication materials) types were defined, namely digital flyers, digital brochures, audios, videos, websites/webpages, social media, mobile apps, and video games. Social media and mobile apps were only considered if dedicated to the selected topics (see section 2.2.1 ), while institutional accounts (general) or similar were not included.
2.2.2 Topics
Each communication material, based on its content and key messages, was assigned to the following three defined topics, of which two contained sub-topics:
- COVID-19 vaccines : this category includes information about licensed and available COVID-19 vaccines, their underlying data, scientific background, possible side effects, production stages, regulatory status, types of vaccines, Q&As, etc. Information on vaccination appointments (e.g., locations, timelines), travel rules, isolation guidelines, etc. was not included.
-
Clinical trial participation
- Clinical study participation : materials to inform and instruct prospective volunteers about participation in clinical trials and biomedical studies and information on how and where to participate in the studies was included.
- COVID-19 vaccine trials : specific information about vaccine trials for COVID-19. Materials for this topic contain information about volunteering registries, trials on new vaccines to be tested, how to participate, general information for prospective volunteers, etc.
-
Vaccine R&D
- General vaccine R&D : in this sub-category, general information was gathered on definition, how are vaccines developed, the process and stages of general vaccine research, current regulatory requirements, Q&As and composition, among others. Specific information on COVID-19 vaccines was allocated to the topic a. COVID-19 vaccines .
- Pivotal clinical trials : a pivotal clinical trial is a clinical study seeking to demonstrate the safety and efficacy of a new pharmaceutical product to obtain its marketing approval by regulatory authorities. Specific information about pivotal clinical trials (e.g., information regarding clinical trial general definition, phases, Q&A, structure, and implementation, etc.) was labelled in this sub-category.
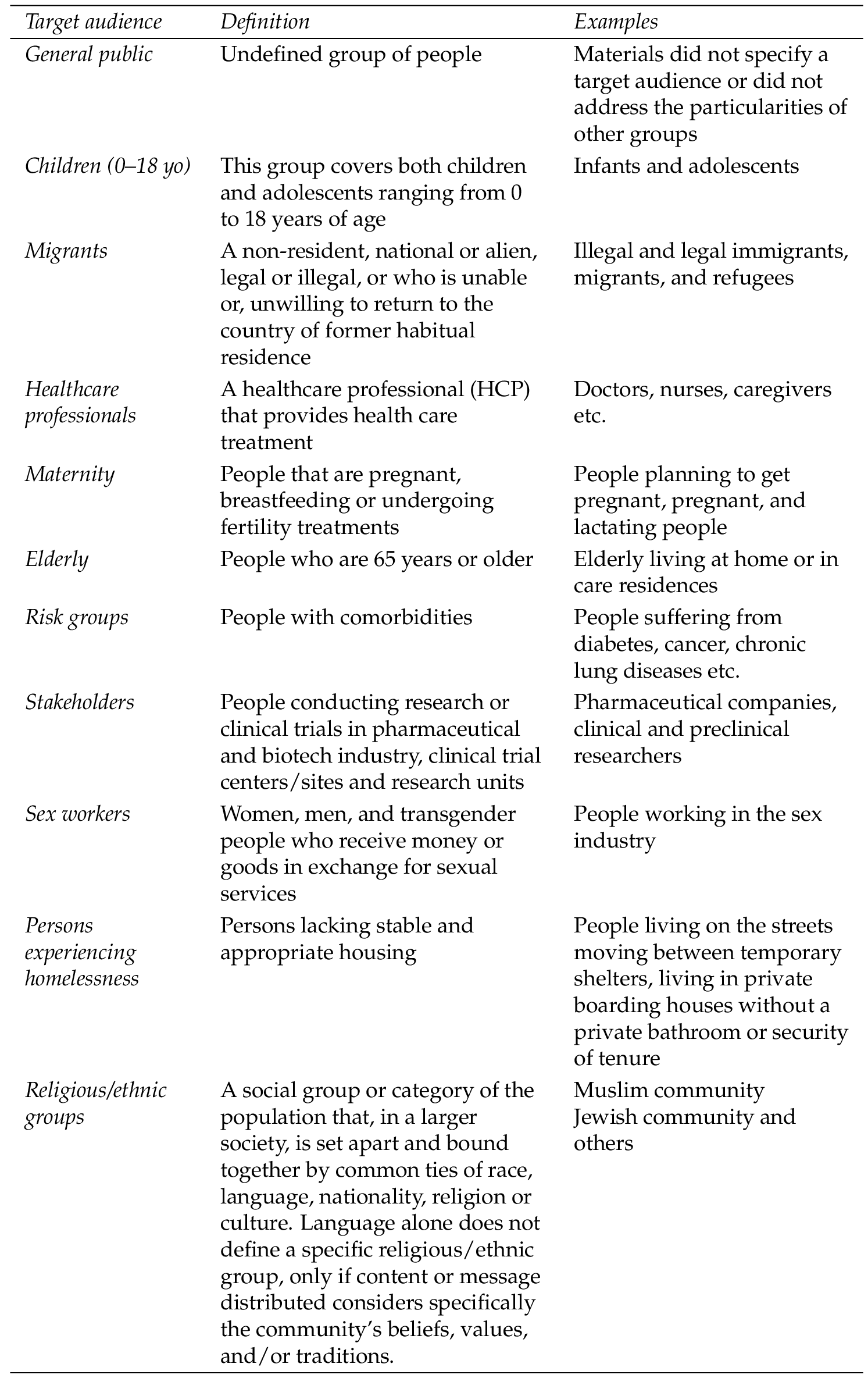
2.2.3 Target audiences
Target audiences were defined into 11 groups, as summarised in Table 1 . If a material was found that referred to two distinct audiences, then it was assigned to both target groups (i.e., it was duplicated and categorised once per target group).
2.2.4 Country
Communication materials were assigned to the country where the issuing institution is located. Multilateral organisations (including NGOs) were assigned to the category EU or Global, depending on their scope of action (i.e., EU-wide or worldwide).
2.2.5 Languages
For each communication material, the language was identified and catalogued.
2.3 Limitations
No data were collected on how often the different online materials were accessed. The current analysis is therefore based on number of publicly available media, not how used they are. Unsurprisingly, the majority of collected material was in the native language of the country and was translated with Google Translator widgets. Therefore, some materials may have been wrongly categorised due to incorrect translation. The number of items of material collected was limited by time as all information was gathered over a specific 3-month period and resources produced after that were not included. The collected information may change, be removed, or links may expire. In many countries age-specific information has been disseminated only once an age-specific COVID-19 vaccination campaign was launched.
Finally, materials collected were limited to online regional, national and European sources, excluding other organisation types (e.g., private companies). It is important to keep in mind that the present inventory did not consider offline sources of information and advertisement campaigns.
3 Results
3.1 COVID-19 vaccine trials-related online information is limited
Most European national governments created dedicated information portals, websites and/or mobile applications for COVID-19 related information (e.g., COVID-19 tracing apps). We specifically looked at these online resources for dedicated sections or materials containing COVID-19 vaccine-related or COVID-19 vaccine trial-related information. A total of 2545 different media materials were identified (see Table 2 ). The majority of materials (84.20%) were related to general information about COVID-19 vaccines , while 9.07% of the material covered Clinical trial participation (with 6.56% for Clinical study participation and 2.51% for COVID-19 vaccine trials ). Fewer resources were found on official websites/apps covering Vaccine R&D (6.72%), including General vaccine R&D (2.87%) and Pivotal clinical trials (3.85%). Thus, dedicated information about COVID-19 vaccine trials was relatively limited compared to more general information about COVID-19 vaccines . Still, the EU institutions produced above-average media dedicated to COVID-19 vaccine trials (15.13%) and Pivotal clinical trials (10.92%) — data not shown.
3.2 Online outreach information does not significantly target underserved communities
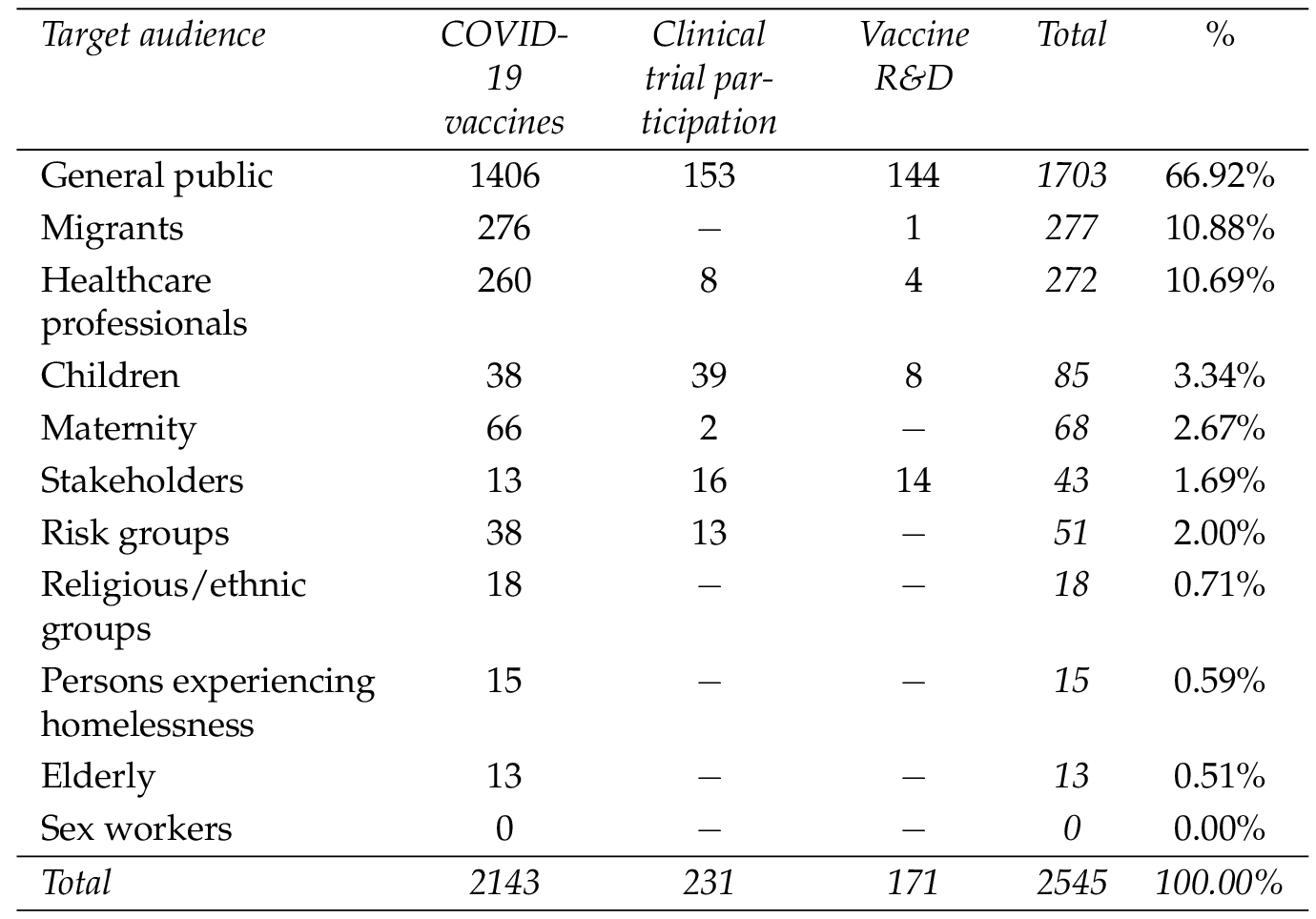
Almost a third of the online resources targeted specific minority audiences or population groups whereas two thirds (66.92%) targeted the General public (Table 3 ). Migrants were the second largest group (after the General public ), targeted in 10.88% of materials collected, while a comparable number (10.69%) was intended for Healthcare professionals . Groups like Children were less often addressed (3.34%), followed by Maternity (2.67%). Interestingly, Risk groups (i.e., people with comorbidities) were targeted only by 2.00% of the media.
Another high-risk group for COVID-19, namely the Elderly , were only targeted in 0.51% of online materials. However, this group represents between 9.10% and 23.20% (median 19.90%) [Eurostat, 2020c ] of the population in the countries investigated. Only four countries (Switzerland, Poland, Spain, and Sweden) produced online media specifically targeting the Elderly group. Similarly, only 3.34% of materials targeted Children , even though they represent between 13.00% and 23.10% (median: 15.70%) of the population of the countries analysed [Eurostat, 2020b ]. 16 countries out of 38, produced materials targeting Children .
Migrants were targeted in 10.88% of media items found, while the median percentage of this group among the populations in the examined countries only constituted 8.45% [Eurostat, 2020a ]. However, the relatively high number of media items for this group was created by only four countries (Norway, U.K., Switzerland, and Greece) and EU institutions. This reveals a striking imbalance among countries since no materials were found online specifically targeting this group from most countries. Healthcare professionals were targeted by 10.69% of media found. In comparison, they represent only 0.25% to 3.89% (median 1.32%) of the populations in the countries assessed [Eurostat, 2018a , 2018b ]. Nevertheless, over half of the countries (22 out of 38) produced media dedicated to this target group. The scarcity of materials devoted to some groups becomes even more apparent when analysing each topic individually. For COVID-19 vaccine trials , no information was found targeting Migrants, Religious/ethnic groups, Persons experiencing homelessness and Elderly . EU institutions covered the highest number of different audiences, with specific media targeting 8 out of 10 defined groups. The United Kingdom (7 out of 10), Poland, Switzerland and Iceland also targeted a high number (6 out of 10) of distinct audience segments. At the same time, countries such as Portugal, Malta, Lithuania, and Romania released materials solely targeting the General public .
3.3 Bulk of media in English, French, and German
From the 2545 online media material list, we identified material in 90 different languages. However, more than a third (36%) is available in only three languages: English, French, and German with 20%, 10% and 6% respectively. Many countries (27 out of 38) created materials in English and their local language, therefore making English the most dominant language overall.
This inventory analysis made it possible to identify how many languages each country made materials available and took care of their non-native national language speakers. Norway made its media/materials available in 48 languages. It had by far the vastest coverage of languages, followed by United Kingdom who covered 37 languages, and by Belgium, who translated its media/materials into 30 different languages, while the EU ensured availability of its media in 27 languages.
3.4 Flyers, websites and brochures were the preferred formats to disseminate information
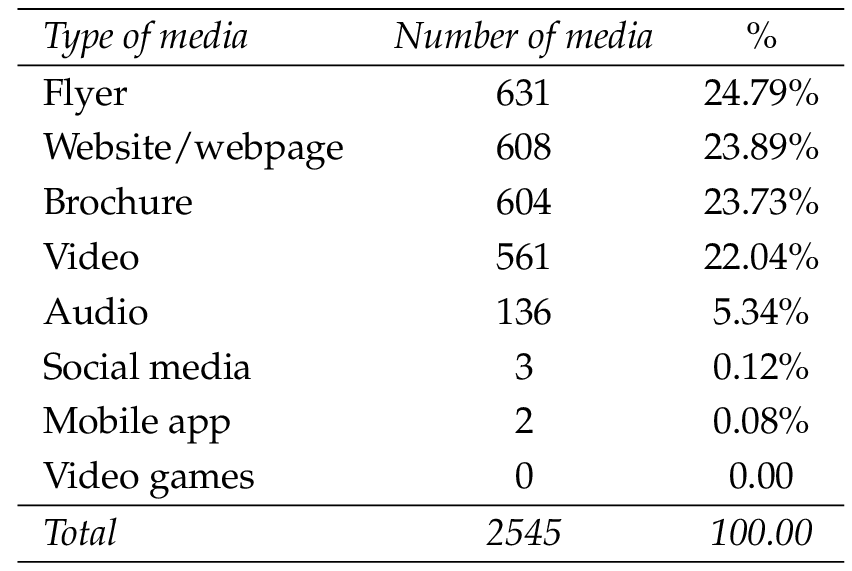
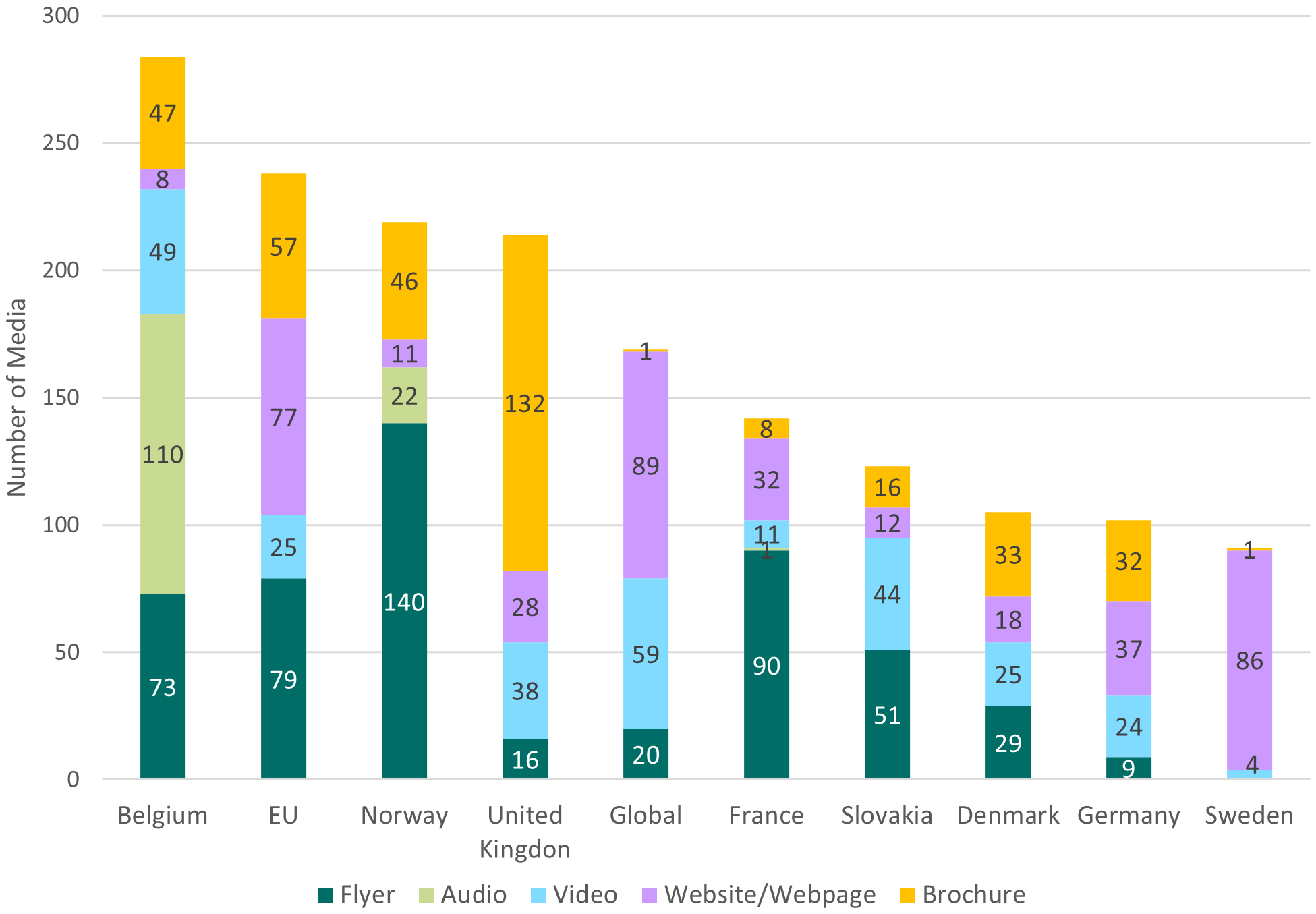
This analysis included eight different media formats, and overall, the most widely used format was a flyer (24.79%), shortly followed by a website/webpage (23.89%) and brochures (23.73%). In contrast, few dedicated social media and mobile apps were found and no video games (Table 4 ). However, there were significant national differences in the use of different formats (Figure 1 ).
Each country (including EU) produced media in several different formats. From the eight formats considered (i.e., Flyers, Brochures, Videos, Video games, Audios, Websites, Social media channels and Mobile applications), Belgium used the largest variety of different formats (5), comprising 47 brochures, 8 links to different webpages, 49 videos, 110 different audios and 73 flyers. Norway created content in four formats (e.g., audio, flyers, brochures and websites) and the United Kingdom produced flyers, brochures and websites. At the same time, the European Union institutions, produced the second-largest number of media, with 79 flyers and 57 brochures and additional 77 webpages and 25 explicative videos, but no audios were found (Figure 1 ).
3.5 National vaccination rates do not correlate with online media intensity
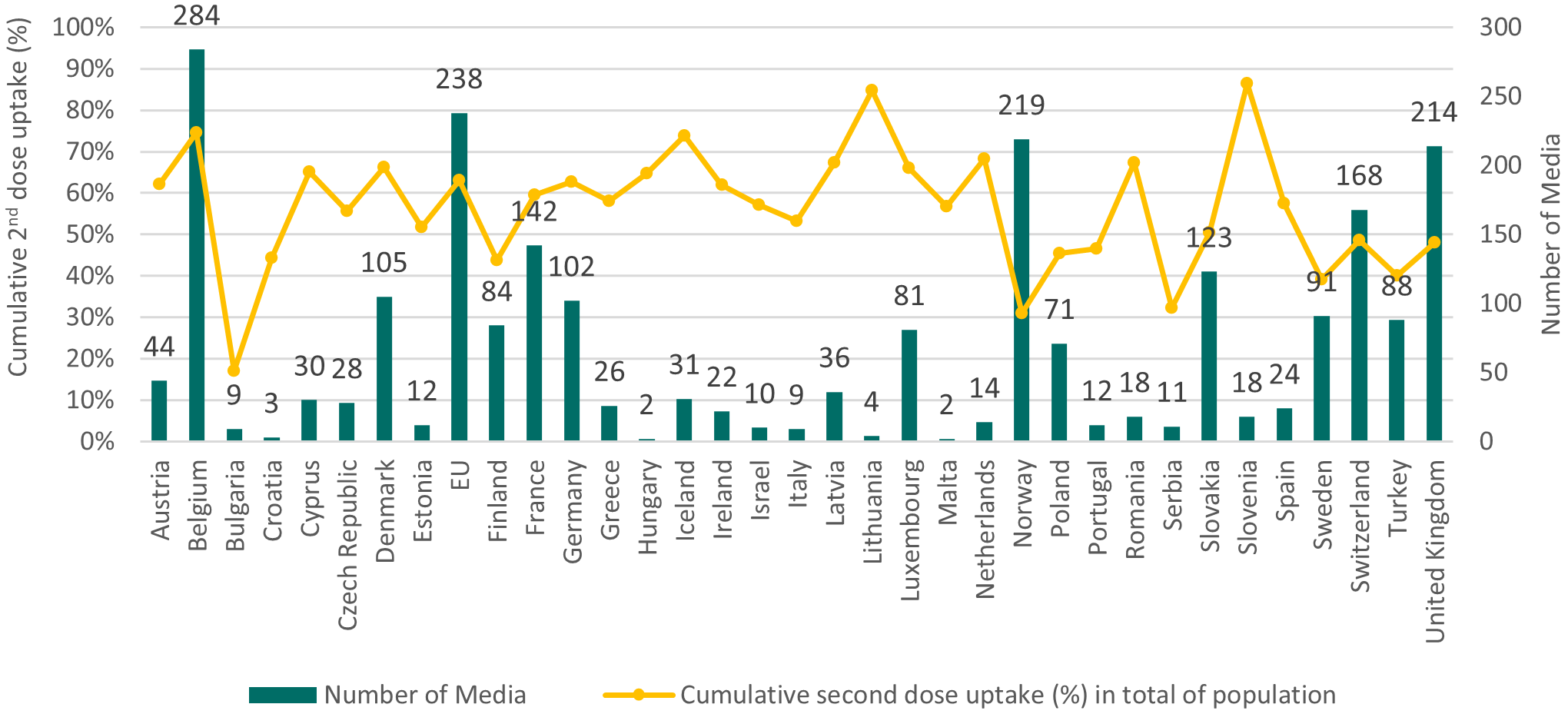
The number of online media made available by official sources in different countries was highly variable. Some countries produced a high number of different materials, while others produced significantly less. Overall, the largest number of materials was produced by Belgium (284), EU (238), Norway (219), U.K. (214) and Switzerland (168), while the smallest number of materials was produced by Croatia (3), Malta (2) and Hungary (2). The number of different materials was therefore not related to the size of the country, with relatively small countries like Belgium, Norway and Switzerland making hundreds of media available and relatively large countries like Spain and Romania producing relatively few different online materials. To assess the impact of the number of available online media on vaccination rates, the number of online media was correlated with national vaccination uptake (cumulative second dose uptake (%) in total of population) at the time of the information collection (Figure 2 ). However, no correlation ( ) was observed between the number of media made available by a country and the same country’s vaccination rate.
3.6 COVID-19 Vaccine Trials information does not depend on clinical trials conducted
Finally, we examined whether the number of vaccine trials in a country correlated with the amount of information material that was available. It could be hypothesised that countries with more COVID-19 vaccine trial activities would also be encouraged to produce more information material about clinical trials. However, we found little or no correlation between the number of vaccine trials and the number of online information material in a country. While Germany conducted the highest number (20) of vaccine trials [European Medicines Agency, 2021 ], it produced only five materials/media about COVID-19 vaccine trials . Austria and U.K., both with nine trials, had produced none and two materials, respectively. On the other hand, France conducted only eight trials, but no less than 18 materials/media on this topic. Overall, the amount of specific material on COVID-19 vaccine trials was limited and concentrated in a few countries. Considerably more countries produced information on Clinical trials participation (16 countries including EU) and Pivotal clinical trials (19 countries including EU).
4 Discussion
We have compiled an inventory of official online resources with information materials about COVID-19 vaccines and vaccine trials. The inventory comprises over 2500 entries from across Europe and neighbouring countries. By reviewing the content, topic area, targeted audience, type of media, language and country of origin of the different media, we analysed the use of online communication by European authorities to inform about COVID-19 vaccines and vaccine trials. Most of the information material (66.92%) was directed towards the General public , whereas about a third was targeting specific population groups. As expected, most of the information was dedicated to general information about COVID-19 vaccines (84.20%).
A modest 2.51% of the material collected was focused on COVID-19 vaccine trials , and this material was furthermore concentrated in only four languages: English, French, German and Czech. Additionally, all the material (64 entries) covering this topic were websites, while other media types were not used. Literacy bias is an important, and often hidden, underlying barrier to achieving diversity in clinical research [Khan, Asif, & Jaffery, 2021 ; Lo, 2007 ]. At least 6 countries had ongoing clinical trials in children or adolescents during the period of analysis, and a fair amount of the total online media information on Clinical trial participation was specifically targeting Children (39 out of 231, corresponding to 17%). During the data collection period, there was substantial debate about the necessity to vaccinate children. This controversy has partially persisted, and European countries continue to have different vaccine recommendations for children. It may also have triggered an urge to develop a relatively high number of online information material for children. Contrary to this, little or no information about Clinical trial participation was identified for other key target groups such as Maternity , or Risk groups . Similarly, no targeted information for the Elderly (65+) on COVID-19 vaccine trials was found, although this is the largest risk group for severe COVID-19 outcomes. The surprising absence of online targeted material for this group might be partially explained by the fact that online media may not be the preferred information channel for this target audience. Instead, this group may rely primarily on other information sources such as primary healthcare and traditional media like newspapers, radio or television. In addition, information and recruitment may have been disseminated through other sources, such as, universities and hospitals, where vaccine trials were conducted for specific groups (e.g., Children , Maternity , or Risk group ), which were not the main sources targeted. This may contribute to the lack of representation of these groups in the current clinical trial landscape [Flores et al., 2021 ; Heller et al., 2014 ].
The amount of material produced per country and target group does not generally match the proportion of those target groups in their national population. Similarly, media production among countries differed greatly. While some countries produced a large number of media targeting almost all the defined audiences, most of the countries produced just a small number of materials targeting mainly General public and Healthcare professionals , leaving the rest of the groups behind. For example, while the category Migrants was the second-largest group targeted in the overall media/materials, these materials were produced only by five constituencies (Norway, U.K., Switzerland and Greece and the EU) out of 38 countries.
The shortage of information related to clinical trials (including COVID-19 vaccine trials ) and dedicated to a particular audience (e.g., Elderly ) may be because pharmaceutical companies, and not official public sources, are often responsible for conducting vaccine trials and recruiting volunteers for their trials. Hence, there may be information available for those audiences online; however, these materials are not produced by official public, international, national, and multilateral organisations. This is nevertheless an important shortcoming as it means that no information is easily accessible from sources with no commercial interests. Our finding that countries with many ongoing clinical trials did not produce markedly more official information materials about clinical trials or trial participation, seems to corroborate this. Centralised official public sources without commercial interests, such as the VACCELERATE Volunteer Registry, as an active single-entry point for European residents interested in clinical trials participation, aim to address some of the shortcomings in information and accessibility to vaccine trials [Salmanton-Garcia et al., 2022 ].
Interestingly, we found no apparent correlation between the amount of media/materials produced and the level of vaccination uptake. Countries with fewer official online materials had similar or even higher vaccination rates as countries with substantial number of available official online materials. This could indicate that a country’s vaccination rate depends much more on vaccine availability and organisation of vaccine distribution than on access to online information about them. However, the present analysis cannot reveal the overall impact of communication intensity on vaccination uptake as it is restricted to online media. The success of vaccination campaigns may thus depend heavily on other channels and resources allocated to dissemination. Online official sources may not be the primary or single source of information for many groups. National news outlets, social media [Cuello-Garcia, Pérez-Gaxiola, & van Amelsvoort, 2020 ; Li, Bailey, Huynh, & Chan, 2020 ], influencers [Mirbabaie, Bunker, Stieglitz, Marx, & Ehnis, 2020 ] general practitioners, friends and family members, colleagues and peers [Geçer, Yıldırım, & Akgül, 2022 ] may have a higher impact on some population groups when compared to directly accessing official online. Moreover, our data only compared the online media intensity with vaccination rates during the early phase of mass vaccination in Europe. Most European countries began large scale roll-out of COVID-19 vaccines in early 2021, and we collected data on online media intensity from the first six months of the vaccination roll-out. It is therefore possible that sustained and high level of online media intensity will correlate with high levels of vaccination in the long run.
5 Conclusion
Adequate online communication by official authorities on COVID-19 vaccine trials (including trial participation) was lacking in many parts of Europe, particularly targeting specific under-represented groups. Countries within Europe perform very differently regarding amount, type, inclusiveness, diversity of materials and particular audiences (un)targeted. The EU institutions have partially harmonised and levelled the field both in terms of topics and diversity of materials and audiences addressed in the effort towards tackling the pandemic. Remarkably, we did not find any association between vaccination rates in the general population and the information made available online by official sources. Countries with extensive online information material did not generally appear to have vaccination rates that were higher than countries with little or no online information. Nevertheless, such association may exist in specific population groups or may manifest itself upon high and long-term online media intensity. Concerted efforts must therefore be pursued to ensure the information reaches the intended audiences, likely requiring cooperation and alignment at the European and national levels to maximise resources, impact and reach of such materials. This is of great importance, especially when addressing distinct groups with singular social, economic and cultural characteristics, which tend to be underrepresented in clinical research at large and particularly in clinical trials. Such actions will be key in strengthening sustainable public health capacities and our ability to tackle future outbreaks.
Acknowledgments
This project has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement No 101037867 (VACCELERATE). This communication reflects the author’s views only, and the European Union cannot be held responsible for any use which may be made of the information contained therein. The research leading to these results was conducted as part of the VACCELERATE consortium. For further information please refer to https://vaccelerate.eu/ .
6 Access to source material
This inventory that was established as part of the present study is made publicly available online for the purpose of knowledge sharing with any interested party. It can be accessed from the VACCELERATE official website ( https://vaccelerate.eu/ ) or from the European Vaccine Initiative website ( https://www.euvaccine.eu/vaccelerate-inventory-report-covid19-vaccine-trials ). The inventory’s interface has been completely built in English in order to make it easily accessible and understandable. All material and project titles have been translated into English.
References
-
Cuello-Garcia, C., Pérez-Gaxiola, G., & van Amelsvoort, L. (2020). Social media can have an impact on how we manage and investigate the COVID-19 pandemic. Journal of Clinical Epidemiology 127 , 198–201. doi: 10.1016/j.jclinepi.2020.06.028
-
European Centre for Disease Prevention and Control (2021). COVID-19 Vaccine Tracker. Retrieved from https://vaccinetracker.ecdc.europa.eu/public/extensions/COVID-19/vaccine-tracker.html
-
European Medicines Agency (2021). EU Clinical Trials Register. Retrieved from https://www.clinicaltrialsregister.eu/ctr-search/search
-
Eurostat (2018a). Practising nurses and caring professionals, 2018. Retrieved from https://ec.europa.eu/eurostat/statistics-explained/index.php?title=File:Practising_nurses_and_caring_professionals,_2018_Health20.png
-
Eurostat (2018b). Practising physicians. Retrieved from https://ec.europa.eu/eurostat/databrowser/view/tps00044/default/table?lang=en
-
Eurostat (2020a). Migration and migrant population statistics. Retrieved from https://ec.europa.eu/eurostat/statistics-explained/index.php?title=Migration_and_migrant_population_statistics#Migrant_population:_23_million_non-EU_citizens_living_in_the_EU_on_1_January_2020
-
Eurostat (2020b). Population by age group (0–14 years). Retrieved from https://ec.europa.eu/eurostat/databrowser/view/tps00010/default/table?lang=en
-
Eurostat (2020c). Proportion of population aged 65 and over. Retrieved from https://ec.europa.eu/eurostat/databrowser/view/tps00028/default/table?lang=en
-
Fazel, M., Puntis, S., White, S. R., Townsend, A., Mansfield, K. L., Viner, R., … Freeman, D. (2021). Willingness of children and adolescents to have a COVID-19 vaccination: results of a large whole schools survey in England. eClinicalMedicine 40 , 101144. doi: 10.1016/j.eclinm.2021.101144
-
Flores, L. E., Frontera, W. R., Andrasik, M. P., del Rio, C., Mondríguez-González, A., Price, S. A., … Silver, J. K. (2021). Assessment of the inclusion of racial/ethnic minority, female, and older individuals in vaccine clinical trials. JAMA Network Open 4 (2), e2037640. doi: 10.1001/jamanetworkopen.2020.37640
-
Garg, I., Hanif, H., Javed, N., Abbas, R., Mirza, S., Javaid, M. A., … Sheikh, A. B. (2021). COVID-19 vaccine hesitancy in the LGBTQ+ population: a systematic review. Infectious Disease Reports 13 (4), 872–887. doi: 10.3390/idr13040079
-
Geçer, E., Yıldırım, M., & Akgül, Ö. (2022). Sources of information in times of health crisis: evidence from Turkey during COVID-19. Journal of Public Health 30 (5), 1113–1119. doi: 10.1007/s10389-020-01393-x
-
Heller, C., Balls-Berry, J. E., Nery, J. D., Erwin, P. J., Littleton, D., Kim, M., & Kuo, W. P. (2014). Strategies addressing barriers to clinical trial enrollment of underrepresented populations: a systematic review. Contemporary Clinical Trials 39 (2), 169–182. doi: 10.1016/j.cct.2014.08.004
-
IGI Global (n.d.). What is Underserved population. Retrieved August 4, 2021, from https://www.igi-global.com/dictionary/underserved-population/30907
-
Israel Ministry of Health (2021). COVID-19 Data Tracker — Overview. Retrieved from https://datadashboard.health.gov.il/COVID-19/general?utm_source=go.gov.il&utm_medium=referral
-
Khan, S., Asif, A., & Jaffery, A. E. (2021). Language in a time of COVID-19: literacy bias ethnic minorities face during COVID-19 from online information in the UK. Journal of Racial and Ethnic Health Disparities 8 (5), 1242–1248. doi: 10.1007/s40615-020-00883-8
-
Li, H. O.-Y., Bailey, A., Huynh, D., & Chan, J. (2020). YouTube as a source of information on COVID-19: a pandemic of misinformation? BMJ Global Health 5 (5), e002604. doi: 10.1136/bmjgh-2020-002604
-
Lo, B. (2007). Addressing ethical issues. In S. B. Hulley, S. R. Cummings, W. S. Browner, D. G. Grady, & T. B. Newman (Eds.), Designing clinical research (3rd ed., pp. 225–239). Philadelphia, PA, U.S.A.: Lippincott Williams & Wilkins.
-
Longchamps, C., Ducarroz, S., Crouzet, L., Vignier, N., Pourtau, L., Allaire, C., … Melchior, M. (2021). COVID-19 vaccine hesitancy among persons living in homeless shelters in France. Vaccine 39 (25), 3315–3318. doi: 10.1016/j.vaccine.2021.05.012
-
Mirbabaie, M., Bunker, D., Stieglitz, S., Marx, J., & Ehnis, C. (2020). Social media in times of crisis: learning from Hurricane Harvey for the coronavirus disease 2019 pandemic response. Journal of Information Technology 35 (3), 195–213. doi: 10.1177/0268396220929258
-
Republic of Turkey Ministry of Health (2021). COVID-19 Vaccination Information Platform. Retrieved from https://covid19asi.saglik.gov.tr/?_Dil=2
-
Salmanton-García, J., Stewart, F. A., Heringer, S., Koniordou, M., Álvarez-Barco, E., Argyropoulos, C. D., … the VACCELERATE consortium (2022). VACCELERATE: Volunteer Registry: a European study participant database to facilitate clinical trial enrolment. Vaccine 40 (31), 4090–4097. doi: 10.1016/j.vaccine.2022.05.022
-
Scherer, A. M., Gedlinske, A. M., Parker, A. M., Gidengil, C. A., Askelson, N. M., Petersen, C. A., … Lindley, M. C. (2022). Acceptability of adolescent COVID-19 vaccination among adolescents and parents of adolescents — United States, April 15–23, 2021. MMWR. Morbidity and Mortality Weekly Report 70 (28), 997–1003. doi: 10.15585/mmwr.mm7028e1
-
UK Health Security Agency (2021). Coronavirus (COVID-19) in the UK. Vaccinations in United Kingdom. Retrieved from https://coronavirus.data.gov.uk/details/vaccinations
Authors
Catarina Luís, European Vaccine Initiative. E-mail: communication@euvaccine.eu .
Veronica Romina Di Marzo, European Vaccine Initiative. E-mail: romidimarzo@gmail.com .
Mandeep Kaur, European Vaccine Initiative. E-mail: mandeep.kaur@euvaccine.eu .
Christos D. Argyropoulos, School of Medicine, European University Cyprus (EUC). E-mail: C.Argyropoulos@research.euc.ac.cy .
Declan Devane, National University of Ireland Galway, Evidence Synthesis Ireland & Cochrane Ireland. E-mail: declan.devane@nuigalway.ie .
Fiona A. Stewart, University of Cologne, Faculty of Medicine and University Hospital Cologne, Department Internal Medicine, Excellence Center for Medical Mycology (ECMM), Cologne, Germany University of Cologne, Faculty of Medicine and University Hospital Cologne, Cologne Excellence Cluster on Cellular Stress Responses in Aging-Associated Diseases (CECAD), Cologne, Germany (UHC). E-mail: fiona.stewart@uk-koeln.de .
George Antoniou, Department of Information Systems & Operations, European University Cyprus (EUC). E-mail: g.antoniou@euc.ac.cy .
Greet Hendrickx, Universiteit Antwerpen, Antwerp, Belgium, Faculty of Medicine and Health Science, VAXINFECTIO, Centre of Evaluation of Vaccination. E-mail: greet.hendrickx@uantwerpen.be .
Helena Hervius Askling, Karolinska Institutet, Department of Medicine/Solna, Division for Infectious Diseases and Karolinska University Hospital. E-mail: helena.hervius.askling@ki.se .
Margot Hellemans, Universiteit Antwerpen, Antwerp, Belgium, Faculty of Medicine and Health Science, VAXINFECTIO, Centre of Evaluation of Vaccination. E-mail: margot.hellemans@uantwerpen.be .
Miriam Cohen, Ministry of Health, Israel. E-mail: miriam.cohen-k@moh.gov.il .
Orly Spivak, Ministry of Health, Israel. E-mail: orlee.f@gmail.com .
Pierre Van Damme, Universiteit Antwerpen, Antwerp, Belgium, Faculty of Medicine and Health Science, VAXINFECTIO, Centre of Evaluation of Vaccination. E-mail: pierre.vandamme@uantwerpen.be .
Rebecca Jane Cox, University of Bergen (UIB). E-mail: rebecca.cox@uib.no .
Sirkka Vene, Karolinska University Hospital. E-mail: sirkka@vene.se .
Sofie Sibia, Karolinska University Hospital. E-mail: Sofie.sibia@regionstockholm.se .
Zoi Dorothea Pana, School of Medicine, European University Cyprus (EUC). E-mail: Z.pana@euc.ac.cy .
Ole F. Olesen, European Vaccine Initiative. E-mail: ole.olesen@euvaccine.eu .

VACCELERATE Consortium is a pan-European network for large-scale clinical vaccine trials to accelerate phase 2 & 3 COVID-19 vaccine trials. It aims to connect European stakeholders involved in vaccine development to provide a single platform for clinical trial design and conduct. Ultimately, VACCELERATE seeks to improve response capability to the current and newly emerging pandemics, and thus to make decisive contributions to emergency preparedness. VACCELERATE was established with support of the European Commission’s European Health Emergency preparedness and Response Authority (HERA).